Mankind has been striving for longevity not only since Lucas Cranach the Elder's "Fountain of Youth" in 1546, but since its very existence. Over time, quacks became scientists and hidden mysticism became visible and verifiable facts. Technical evolution gave research the tools it needed to approach the complexity, the changes in the human body over time. Science has been able to identify, sometimes consciously, sometimes just by chance, in experiments, some adjusting wheels at the level of the cells.
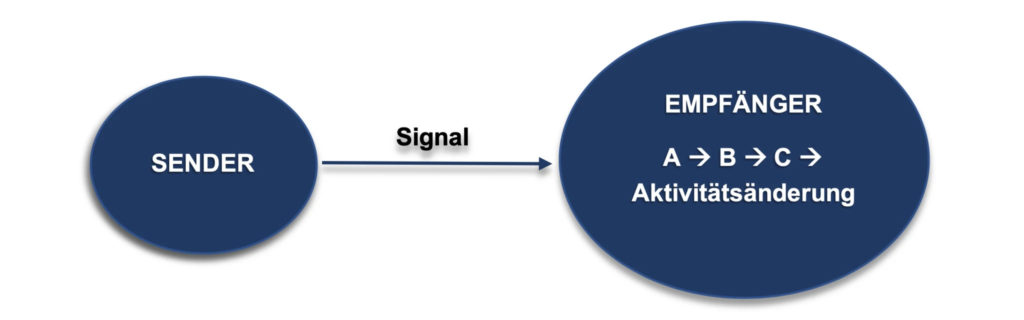
Communication or the exchange of information between cells essentially takes place via small molecules and proteins. As with mobile phone signals, there are one or more transmitter cellsand one or more receiver cells. If the receiver cell registers the signal, chemical messengers pass on the "information" within the cell along special "pathways" and thus lead to a change in the cell's activity. This change in activity can mean, for example, an acceleration or deceleration of work, but it can also mean that the cell suddenly produces completely different products.
Four paths to longevity
With regard to ageing and longevity, four "pathways" have emerged on the molecular playing field:
- mTOR
- AMPK
- Sirtuins
- NF-kB
The discovery of these four pathways has enabled scientists to fundamentally change our understanding of aging. The presentation of these signaling pathways as pathways suggests that they are independent marches through the fountain of youth. However, this is a misconception due to the way they are formulated. mTOR, AMPK, sirtuins, and NF-kB are interrelated in many ways and are sometimes mutually dependent. Nevertheless, for ease of comprehension, let us take the paths toward longevity one at a time. Let's get started.
mTOR - Rapamycin, mice and fasting
mTOR (mechanistic target of rapamycin) is a protein found in all mammals including humans that functions as a signaling hub in the cell. This means that mTOR senses and integrates many signals to control reactions such as cell growth and division, cell death, and inflammation. If the regulation of mTOR does not function, this can have a negative impact on longevity and health.
Both too much activity and too little proved to be detrimental. Overfunction of the mTOR pathway was found in cardiovascular diseases, cancer, obesity or diabetes. Too low mTOR function weakens our immune system. This can be exploited medically with drugs such as rapamycin, which inhibit mTOR. For example, if someone receives a liver transplant, the immune system must be weakened so that it does not reject the new "foreign" liver.
In addition, rapamycin is the only drug to date that has consistently led to life extension in studies on mice. However, these were kept in a germ-free environment and therefore did not rely on a strong immune system. Nevertheless, the results are encouraging, as inhibition of mTOR activity attenuated age-related diseases such as diabetes, cancer, and heart disease. This interesting approach has already been taken up by researchers, and potential anti-aging therapies based on mTOR have thus come within reach.
We can also contribute to signalling pathway balance independently of possible medication. The golden mean of mTOR function can be achieved by:
- a healthy lifestyle with sufficient exercise,
- occasional fasting episodes
- and the reduction of animal proteins in our meals.
AMPK - Metformin, free radicals and mTOR inhibition
What is true for coexistence is also true on a smaller scale for the cell. Energy metabolism in balance, improved stress resistance and skilled housekeeping are all features of improved healthspan and extended lifespan. The AMPK(adenosine monophosphate-activated protein kinase) signaling pathway is involved in regulating all of these features and may additionally inhibit the mTOR signaling pathway we learned about earlier. As we know, this can have positive effects for us.
In the body, AMPK ensures, for example, that the glucose from our food is converted into energy and does not end up in the form of fat on our hips or bellies. Researchers are exploiting this mechanism in the treatment of diabetes. Metformin is approved as a diabetes drug and acts as an AMPK activator in the body. However, due to the broad efficacy of AMPK, the drug is believed to have radiance far beyond diabetes. The potential for this is currently being investigated in several studies.
We know from various research efforts that the responsiveness of AMPK decreases with age - unfortunately. This loss impairs metabolic regulation and increases the number of free radicals in the body. These age-related changes then cause metabolic disorders and mild inflammation in the body's cells. As a result, we are more likely to develop diabetes or cardiovascular disease.
Fortunately, each of us can contribute to longevity via AMPK to better maintain AMPK functionality: Intermittent fasting, regular physical exercise and eventual weight loss.
Sirtuins - genes of longevity and NAD+
>Sirtuins is a collective term for a family of seven proteins (SIRT 1 - SIRT 7). We can imagine this protein family roughly like politicians. They decide when a project is to be realized, how it should look, whether several projects should run simultaneously or whether projects should be cancelled. In the case of politicians, at least in theory, these decisions are always made for the good of society or, in the case of sirtuins, for the good of the cell and the organism as a whole.
Because of their important role in the ageing process, they are sometimes referred to as longevity genes. They are involved in cell metabolism and can regulate many cell functions, including DNA repair, inflammatory response, cell cycle or cell death.
However, just as a politician needs voters to put him in the position of making decisions, sirtuins need NAD+ to function. This molecule is found in every cell of the human body and is a major metabolic regulator. Without NAD+, we would die - that's how important NAD+ is.
A healthy lifestyle with physical activity, adequate sleep, and fasting episodes can support the integrity of sirtuin function. In addition, molecules have been discovered that can increase NAD+ levels , providing fuel for sirtuin function.
NF-kB - inflammaging
Nuclear Factor kappa-B(NF-kB) is involved in the regulation of inflammatory processes. Similar to mTOR, here too the dose makes the poison. Inflammation is basically a good thing, because it helps to ward off pathogens and protect us from harmful stimuli. A too low functionality of NF-kB has accordingly negative consequences, because our protective shield is weakened. Overactivation, as is often found in old age, leads to chronic inflammation, so-called inflamm-aging. This hip term stands for inflammation aging in German.
When we think of a classic, local inflammation (aphthae in the mouth or joint inflammation), we usually associate it with pain and a restriction of our well-being. In inflamm-aging, these noticeable signs do not occur. Instead, a subliminal inflammation develops in many cells throughout the body, which generally causes lasting damage to cell function. The therapeutic goal is therefore to slow down the age-related hyperfunction of NF-kB. To this end, a healthy lifestyle with little stress and sufficient rest has also proven beneficial.
Quo vadis?
While chronological age is practically unchangeable, with the exception of falsification of documents, biological age is different. On the one hand, age is an ever-increasing number; on the biological side, it is mostly a decrease in the adaptability of the body. This decrease can now be countered - if one takes the right paths towards longevity.
Salminen, A., Huuskonen, J., Ojala, J., Kauppinen, A., Kaarniranta, K., & Suuronen, T. (2008). Activation of innate immunity system during aging: NF-kB signalling is the molecular culprit of inflamm-aging. Ageing research reviews, 7(2), 83-105. https://www.sciencedirect.com/science/article/pii/S1568163707000566
Zineldeen, D. H., Uranishi, H., & Okamoto, T. (2010). NF-kappa B signature on the aging wall. Current drug metabolism, 11(3), 266-275. https://www.ncbi.nlm.nih.gov/pubmed/20406190
Salminen, A., & Kaarniranta, K. (2012). AMP-activated protein kinase (AMPK) controls the aging process via an integrated signalling network. Ageing research reviews, 11(2), 230-241. https://www.sciencedirect.com/science/article/pii/S1568163711000778?via%3Dihub
Lamming, D. W., Ye, L., Sabatini, D. M., & Baur, J. A. (2013). Rapalogs and mTOR inhibitors as anti-aging therapeutics. The Journal of clinical investigation, 123(3), 980-989. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3582126/
Burkewitz, K., Zhang, Y., & Mair, W. B.. (2014). AMPK at the nexus of energetics and aging. Cell metabolism, 20(1), 10-25. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4287273/
Cartwright, T., Perkins, N. D., & Wilson, C. L. (2016). NFKB1: a suppressor of inflammation, ageing and cancer. The FEBS journal, 283(10), 1812-1822. https://www.ncbi.nlm.nih.gov/pubmed/26663363
Kida, Y., & Goligorsky, M. S. (2016). Sirtuins, cell senescence, and vascular aging. Canadian Journal of Cardiology, 32(5), 634-641. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4848124/
Wątroba, M., Dudek, I., Skoda, M., Stangret, A., Rzodkiewicz, P., & Szukiewicz, D. (2017). Sirtuins, epigenetics and longevity. Ageing research reviews, 40, 11-19. https://www.ncbi.nlm.nih.gov/pubmed/28789901
Weichhart, T. (2018). mTOR as regulator of lifespan, aging, and cellular senescence: a mini-review. Gerontology, 64(2), 127-134. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6089343/
Stallone, G., Infante, B., Prisciandaro, C., & Grandaliano, G. (2019). mtor and aging: An old fashioned dress. International journal of molecular sciences, 20(11), 2774. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6600378/