Phospholipids are a familiar term, especially to chemists. They are a special class of lipids (fats). What makes these small molecules special is the fact that they consist of a hydrophilic ("water-loving") head and a hydrophobic ("water-repellent") tail. Experts affectionately refer to this combination as "amphiphilic." From a chemical point of view, the head is composed of a phosphate group and an alcohol group, while long hydrocarbon chains form the two tails.
However, apart from detailed knowledge, each and every one of us has already made the acquaintance of phospholipids. The molecules are found in many foods, especially in fatty foods such as eggs, dairy products, meat and fish. They are also found in food supplements and medicines.
The cells of many living organisms (humans, animals, plants) have taken advantage of this special structure. Specifically, we are talking about the cell membrane, where phospholipids, along with glycolipids and cholesterol, are jointly responsible for its structure. In principle, an aqueous environment prevails in our body. When phospholipids now come into contact with it, the hydrophilic heads turn toward the water and the fatty tails turn away from the water. As a result, the hydrophobic tails cluster inward and the characteristic phospholipid bilayer forms.
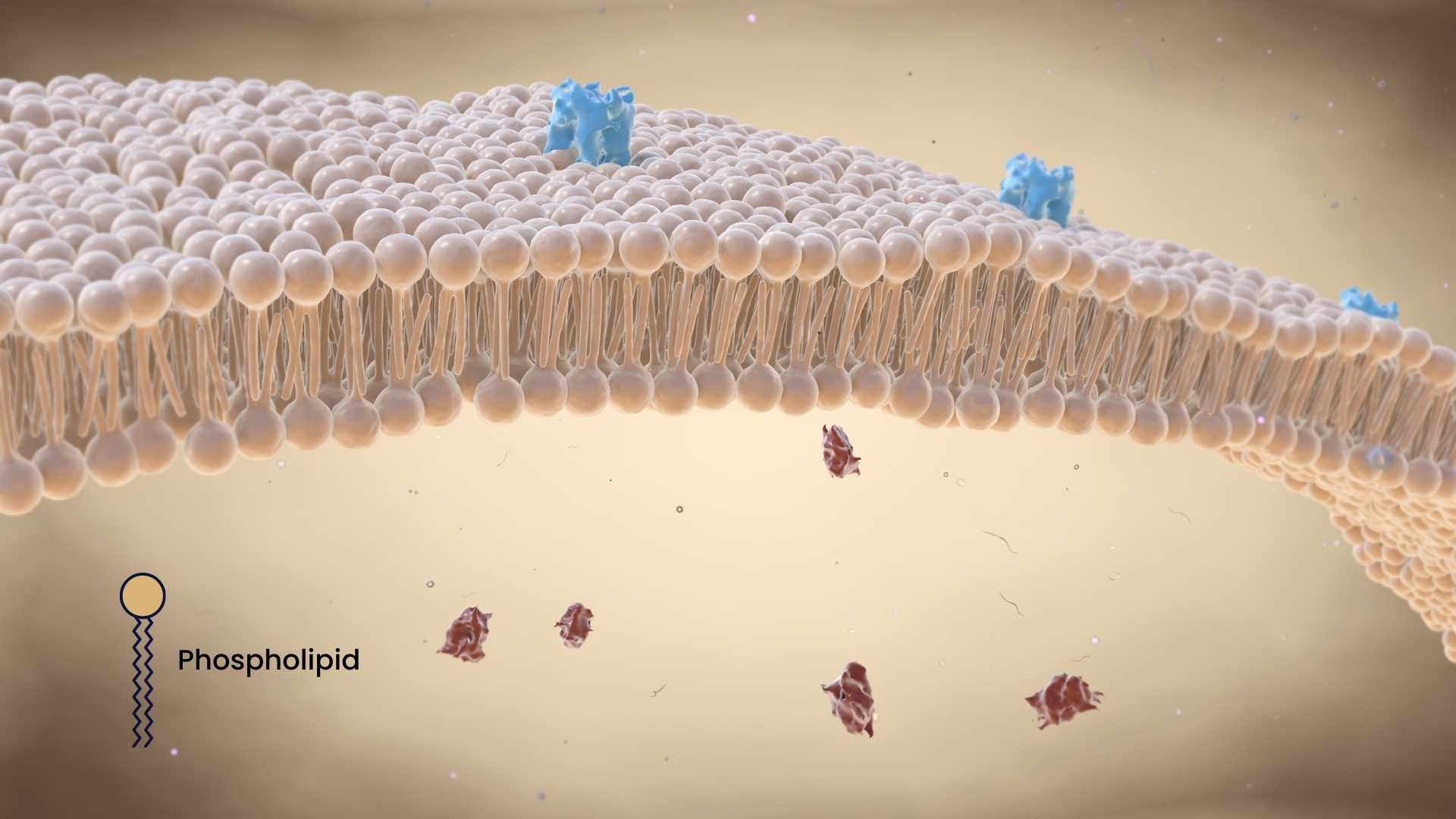
What are the functions of phospholipids in the cell?
First and foremost, phospholipids form the basis for cell membranes and thus maintain the structure of the cells. They also perform a very important selective barrier function, separating the inside of the cell from the outside and preventing the penetration of unwanted substances. At the same time, however, the phospholipid bilayer in the cell membrane allows the import and export of necessary molecules and ions. However, the molecules also have competencies beyond security tasks.
Namely, they serve as precursor molecules for signaling molecules such as inositol trisphosphate and diacylglycerol. As a result, they play an essential role in signal transduction . In adipose tissue, phospholipids can even serve as energy stores.
Where are phospholipids used?
The special chemical properties make the small molecules a highly sought-after component. Whether in the food industry, medicine, cosmetics or biotechnology - phospholipids have a hand in everything.
In the food industry, they are often used as emulsifiers to mix and stabilize water and oil components in foods. This is necessary, for example, in the production of margarine, chocolate, ice cream, baked goods and many other foods.
From a medical point of view, phospholipids are relevant as components of food supplements or drugs. This refers to liposomal mixtures that improve the efficacy and bioavailability of active ingredients. What effect this use can have, you can see at the example of Quercesome. MoleQlar's specially formulated quercetin C complex is 20 times more bioavailable than conventional quercetin powder. The key ingredients are phospholipids from sunflower and the clever combination with natural vitamin C - a bioenhancer of quercetin.
Also at Berbersome the body's berberine absorption capacity is increased 10-fold due to the clever combination with phospholipids!
If they can provide vital substances with easier access to our organism, then this should also work for the skin. This is exactly what the cosmetics industry has been thinking. There, phospholipids are used as moisturizers and carriers in skin care products. They can also be found in lipsticks, shampoos and other cosmetic formulations.
Another major area of application for phospholipids is biotechnology and research. Both areas use the molecules as a component of liposomes for targeted drug release and to improve the stability of cells in cell cultures.
In the cleaning industry, surfactants in detergents and cleaning products contain phospholipids to reduce surface tension and remove dirt.
The best known phospholipids are phosphatidylcholine, phosphatidylethanolamine, phosphatidylinositol and sphingomyelin.









